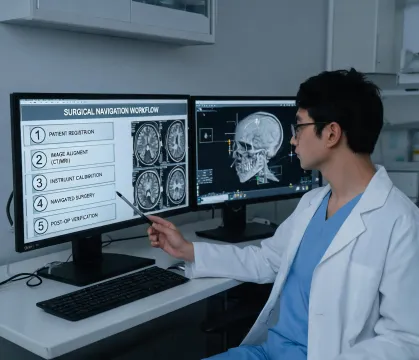
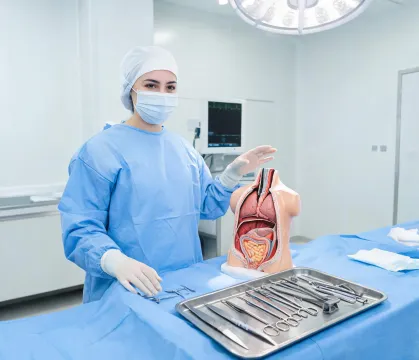
Patient-first engineering
We avoid absolute promises and focus on the device, training, and workflow details that help clinicians make appropriate choices.
QMS discipline
ISO 13485 is treated as a daily operating system: design controls, change records, supplier files, and CAPA thinking shape the work.
Clinician co-design
Surgeons, OR nurses, sterile processing leaders, and biomedical teams influence how resources are organized and how support is delivered.
Regulatory as a skill
We write carefully: FDA cleared when 510(k) applies, CE marked under MDR when appropriate, and no outcome claims without proper support.
Service culture
Engineers and advisors spend time with case support teams so the product conversation includes scheduling, training, and real response paths.
Inclusive hiring
Nuvasive values clinical application specialists, RA/QA professionals, field service talent, and operators who can explain complex topics clearly.